Geo News
What you’re looking at is the story of an infection. Specifically, how the bacterium Citrobacter rodentium spread through hundreds of mice at the University of Auckland’s bioluminescent superbugs lab.
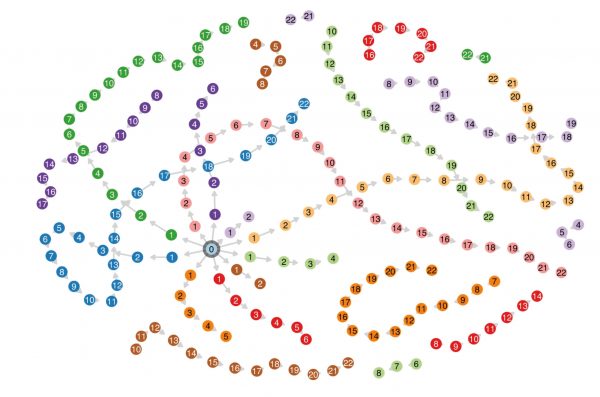
The graphic above shows 10 transmission chains flinging out from a central hub—the original bacterium. The scientists are sure about these transmission chains because they carefully controlled the spread of the disease. To get things started, they inoculated 10 mice with the bacterium (which infects mice, but not humans). Then each of those mice were put in a cage with a healthy mouse. Mice groom one another and eat each other’s poo—unsurprisingly, in most cases the new mouse was quickly infected. Then it was removed and caged with a healthy mouse. And so on, creating distinct chains of transmission. (Breaks in the chains—where a number is missing, and a section of chain floats loose—represent points where a sick mouse failed to infect a healthy one. The scientists would then dig mouse poo from the same chain out of the freezer, and use it to directly infect the stubbornly healthy animal.)
[caption id="attachment_497175" align="alignnone" width="2000"] Researchers exposed mice one by one to a bacterium to build this map of known infection pathways.[/caption]
As the bacterium hopped from mouse to mouse it mutated. What the scientists were interested in, says microbiologist Siouxsie Wiles, who was part of the international team, is whether you could use these “really, really tiny changes”, and the frequency at which they occurred, to trace the path of the infection itself: to figure out, from scratch, which mice got infected first, and who passed the bacteria on to whom.
“What they’ve shown is that actually, you can.”
The diagram on this page is the picture the team were able to piece together using deep gene sequencing and basic information around timing, such as when each mouse tested positive.
[caption id="attachment_497184" align="alignnone" width="2000"] Working backwards using gene sequencing, the researchers tracked tiny mutations in the bacterium to infer the probable chain of transmission. They were able to piece together most mouse-to-mouse transmissions, as well as points where the researchers had to directly infect one mouse using bacteria-ridden poo. Most of the mistakes happen close to the hub, when the bacterium had not mutated much and was harder to track.[/caption]
Wiles explains that it doesn’t matter where the chains sit on the diagrams (that’s just a design algorithm arranging things prettily). The crucial points are that lots of links on each chain are joined up, and they’re more or less in the right order.
“They don’t look identical, but they look pretty damn good considering the data that it had,” Wiles says. Some chains are muddled, but she points out most of those mistakes are happening early on, close to the hub—when the bacterium has not mutated as much, so tracing its evolution is much trickier.
Wiles had been particularly curious about whether the technique would pick up the breaks in the chains, where the transmission had relied on freezer poo. “And it can do that, too, which is quite cool.”
And yes, there’s a COVID application. The findings, published in Nature Communications, have already been used to help figure out that vaccinated people can still get infected, and spread the virus to others.